|
Spatial and Temporal Regulation of Transcription in Development The initial subdivision of an embryo into distinct developmental domains depends on maternally expressed transcription factors that are synthesized, modified, and transported in ways that result in gradients of transcription factor activity. These transcription factor gradients generate discrete domains of expression of a small number of genes in the early embryo. We are focusing on the molecular interactions that allow these maternally encoded transcription factors to generate spatially regulated patterns of transcription.
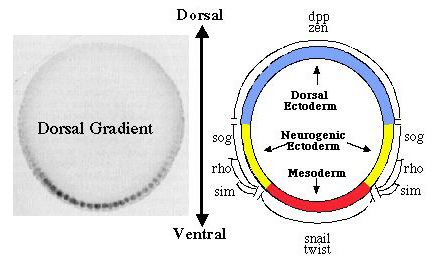
Dorsal functions as both an activator and a repressor of transcription. Early in development, a set of maternally encoded gene products (the maternal dorsoventral patterning system) generates a concentration gradient of the Dorsal protein, a transcription factor that plays a central role in determining the dorsoventral axis of the embryo (Courey and Huang, 1995; Flores-Saaib and Courey, 2000). This gradient results in the spatially regulated expression of genes that are essential for germ layer establishment. For example, since Dorsal is an activator of the twist (twi) gene, twi is only transcribed in the ventrally-situated presumptive mesoderm where Dorsal concentration is high. In contrast, Dorsal is a repressor of the decapentaplegic (dpp) gene, which is therefore only transcribed in the dorsally situated presumptive ectoderm where Dorsal concentration is low. A major goal of our research is to learn what determines whether Dorsal will act as an activator or a repressor of any given target gene. Using a wide variety
of approaches, including protein biochemistry, reverse genetic analysis,
and the generation and analysis of genetic mosaics, we have identified
and extensively analyzed the cis-regulatory elements as well as some
of the protein factors that enable Dorsal to regulate the transcription
of genes such as twi and dpp in the early Drosophila embryo . We have
found that the ventral specific activation of twi depends on ventral-specific
enhancers in the 5' flanking region of the gene, while the ventral repression
of dpp depends on ventral-specific silencers in the second intron of
the gene (Pan
et al., 1991; Pan
and Courey, 1992; Huang
et al., 1993; Pan
et al., 1994). Both the ventral enhancers and the ventral silencers
contain Dorsal binding sites that are essential for the function of
these modules.
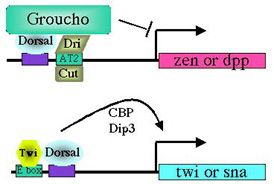
Dorsal binding sites function in a context-dependent manner. What accounts for the ability of Dorsal to activate some genes and repress others? Our findings suggest that it is the context of a Dorsal binding site that determines whether it will mediate activation or repression (Pan and Courey, 1992; Huang et al., 1995; Flores-Saaib et al., 2001). Isolated Dorsal sites mediate activation. However, when Dorsal binding sites occur in the context of certain other regulatory elements they mediate repression. These additional elements are thought toserve as binding sites for proteins that directly or indirectly interact with Dorsal to convert it from an activator into a repressor. The regulatory factors that bind the elements described above are expected to play critical roles in pattern formation. By allowing orsal to function as both an activator and a repressor, these elements should allow the smooth monotonic Dorsal gradient to generate multiple distinct domains of gene activity. Somewhat paradoxically, however, the extensive genetic analysis of Drosophila embryogenesis, which was initiated over 20 years ago by Nüsslein-Volhard and Wieschaus, has failed to reveal the genes that encode these proteins. We have postulated that this is due to genetic pleiotropy as well as genetic redundancy, both of which could obscure the developmental phenotypes of mutations in such genes. We have therefore sought to identify these proteins through biochemical approaches (Huang et al.,1995; Valentine et al., 1998). Critical regulatory factors uncovered in this way include NTF-1, which binds to an element in dpp (the dpp repression element or DRE), as well as Dead Ringer (Dri) and Cut, which bind to an element in zen (the AT2 site). Our analysis of loss of function mutations in the genes encoding Dri and Cut demonstrates that these factors are indeed critical for the function of the AT2 element in zen. As predicted, the roles of the genes encoding these factors in dorsoventral pattern formation was obscured by their pleiotropic roles in multiple developmental processes as well as by the existence of multiple partially redundant control regions within the zen gene. Thus, the discovery of the function of these genes in dorsoventral patterning was only possible because of our extensive application of both biochemical and genetic approaches.
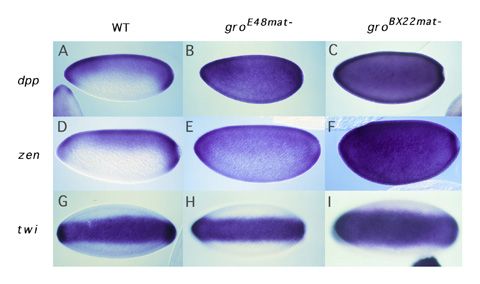
Dorsal-mediated repression requires Groucho. How do factors like Dri and Cut convert Dorsal from an activator to a repressor? Our recent studies on the role of a factor called Groucho in Dorsal-mediated repression provide the probable answer to this question. Groucho is a pleiotropic nuclear protein that has roles in many developmental processes. This pleiotropy makes it impossible to assay the role of the groucho gene in early development by standard genetic techniques. However, using an approach that involved the creation of genetic mosaic flies carrying homozygous groucho germ line clones, we found that Groucho protein was essential for Dorsal-mediated repression (Dubnicoff et al., 1997). As far as we can tell, Groucho does not bind directly to DNA. However, we have recently found that Groucho does bind to both Dorsal and Dri. Furthermore, DNA-bound Dorsal and Dri can cooperatively recruit Groucho to DNA in vitro (Valentine et al., 1998). We thus propose that a platform comprised of Dorsal and DNA-bound "assistant repressors" such as Dri serves to recruit Groucho to the ventral silencer. Mechanism
of Groucho-mediated repression. As a corepressor, Groucho is
recruited to DNA by protein:protein interactions with DNA bound repressor
proteins. Groucho is required for the function of many such DNA-bound
repressors a number of which function during embryogenesis and/or later
developmental stages. Groucho orthologues are present in mammals where
they play roles in such important biological processes as embryonic
pattern formation, neurogenesis, and oncogenesis.
Our recent structure/function
analysis of Groucho is beginning to shed light on the mechanism by which
this protein represses transcription (Courey
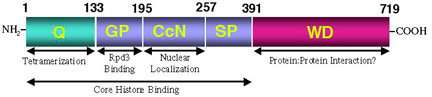
and Jia, 2001). In addition to a WD-repeat domain at its C-terminus,
Groucho contains an evolutionarily conserved region at its N-terminus.
We have found that this conserved N-terminal region mediates the formation
of high order Groucho oligomers (Chen
et al., 1998). Single point mutations within this domain that disrupt
oligomerization also disrupt Groucho-mediated transcriptional repression.
Thus, the ability of Groucho to repress transcription appears to depend
upon the ability of this factor to form high order oligomers. Since
Groucho can repress transcription from long distances, this leads us
to speculate that Groucho mediated repression could involve the oligomerization
of Groucho along the chromosome perhaps to induce long-distance changes
in chromatin structure. Support for the idea that Groucho could oligomerize
along the chromatin fiber comes from our finding that Groucho binds
to histones in vitro (Flores-Saaib
and Courey, 2000).
In
addition to the conserved WD repeat domain and the glutamine-rich tetramerization
domain, Groucho contains another critical domain termed the GP domain
(so-named because of its high content of glycine and proline residues).
We have discovered that this region of Groucho mediates a direct protein:protein
interaction with the histone deacetylase Rpd3 (Chen
et al., 1999). Biochemical assays demonstrate that this interaction
as well as Rpd3 histone deacetylase activity are critical for the ability
of Groucho to repress transcription. Furthermore, we have discovered
a genetic interaction between the genes encoding Groucho and Rpd3 indicating
that the interaction discovered through biochemical means is required
for normal Drosophila embryogenesis. Thus, it appears that Groucho represses
transcription by recruiting chromatin remodeling enzymes such as histone
deacetylases to the template. These enzymes may then modify chromatin
structure altering the accessibility of the template to the transcriptional
machinery.
Efficient
activation by Dorsal requires synergistic interactions with other factors.
In the absence of DNA-bound assistant repressors, Dorsal protein directs
transcriptional activation. Efficient activation requires synergistic
interactions between Dorsal and other sequence specific transcription
factors. For example, genetic studies indicate that certain basic-helix-loop-helix
(bHLH) transcription factors (e.g., the products of twi, daughterless,
and the achaete scute complex) are able to amplify the ventral specific
transcription directed by Dorsal.
Using in vitro transcription
and transient transfection assays, we have found that the Twi bHLH factor
and Dorsal do indeed interact in a synergistic manner (Shirokawa
and Courey, 1997). Deletion analysis of Dorsal indicates that a
region termed the rel homology domain (RHD) is sufficient for synergistic
activation.The RHD is a 300 amino acid domain that has been previously
shown to mediate dimerization, DNA binding, and regulated nuclear import
of Dorsal. To better understand the role of the RHD in transcriptional
control, we carried out an alanine scan mutagenesis of the RHD (Jia
et al., 2002). Two classes of mutations that alter transcriptional
activation without affecting other RHD functions such as DNA binding
and regulated nuclear import were identified. The first class severely
reduces activation in both S2 cells and embryos. In addition, mutant
proteins of this class have lost the ability to repress transcription
strongly suggesting a mechanistic link between activation and repression.
In contrast, the second class increases activation potential under some
conditions, an effect that does not appear to be due to a disruption
in the interaction between Dorsal and its cytoplasmic inhibitor Cactus.
The two classes of mutations are apparently clustered on opposite sides
of the RHD suggesting that the mutations define surfaces for interactions
with distinct proteins. Thus, in addition to tethering Dorsal to cis-regulatory
elements, the RHD plays an active role in transcriptional regulation.
|